Jenny Minigh

Meet our Executive Vice President, Medical Writing
Read More...Rare Disease Clinical Endpoints: Ingenuity Meets Practicality

Discover why the science that brought you to this point can only […]
Read More...CRAs Must Evolve Alongside Oncology Therapeutics
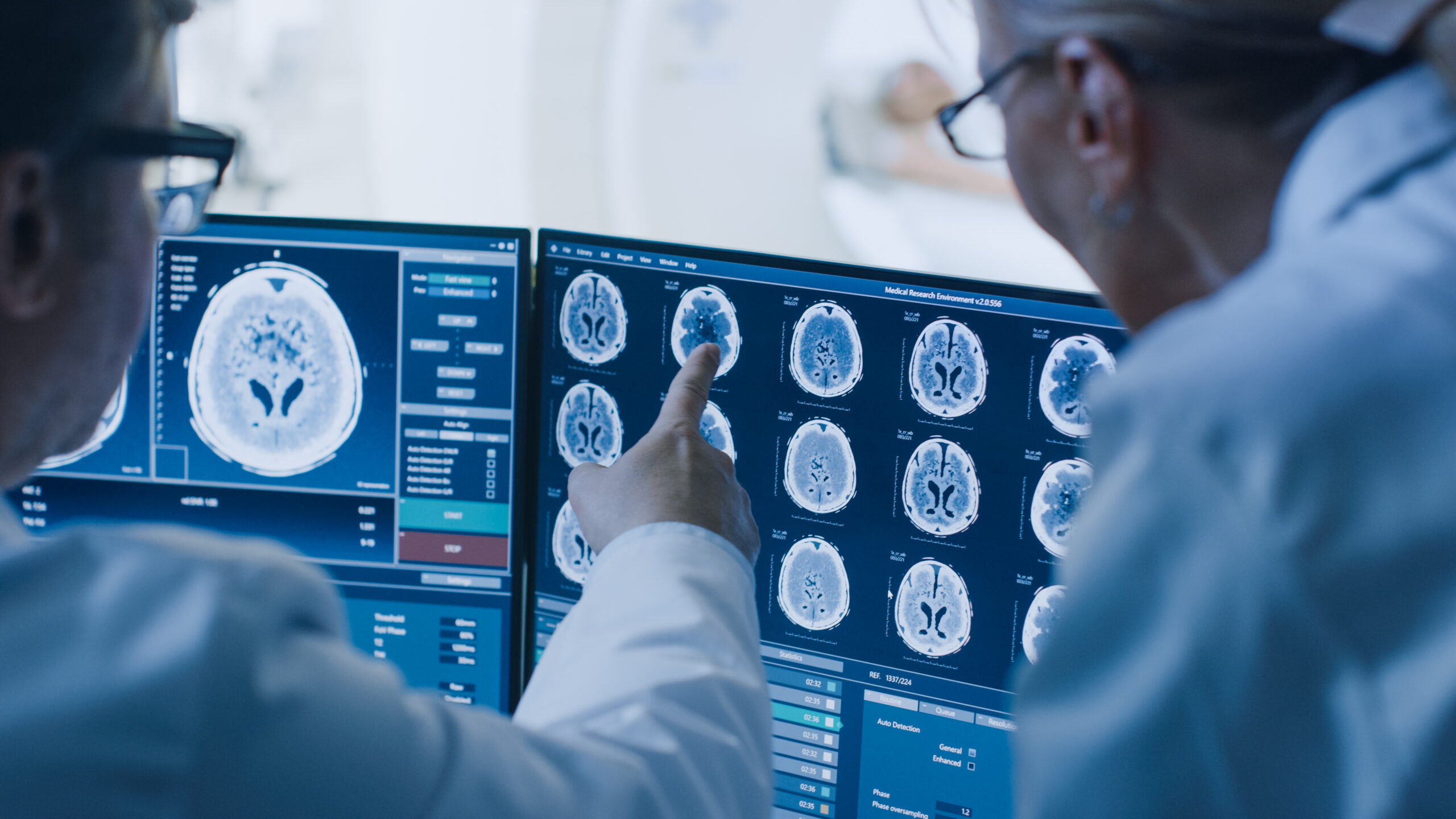
Clinical Research Associates (CRAs) must adapt to the evolving demands of oncology […]
Read More...Crissy Reynolds

Meet our Senior Director, Regulatory Operations
Read More...Raul P. Lima

Meet our Executive Vice President, Clinical Operations
Read More...Karin Rezzonico

Meet our Senior Director, Quality Assurance & Compliance
Read More...Our message to CMOs

For over 12 years, we’ve stood alongside the Chief Medical Officer Summit […]
Read More...FDA Update on ICH E6, E8, QbD and RBM in Clinical Trials

Integrating Quality by Design (QbD) and Risk-Based Monitoring (RBM) approaches in clinical […]
Read More...Revolutionizing Clinical Trials: The Power And Potential Of Risk-Based Monitoring

The article explores the benefits and misconceptions of risk-based monitoring (RBM) in […]
Read More...How Sponsors and Patients Benefit From a Site-Centric Partnership

Site-centric partnerships between sponsors and clinical trial sites can significantly enhance patient […]
Read More...